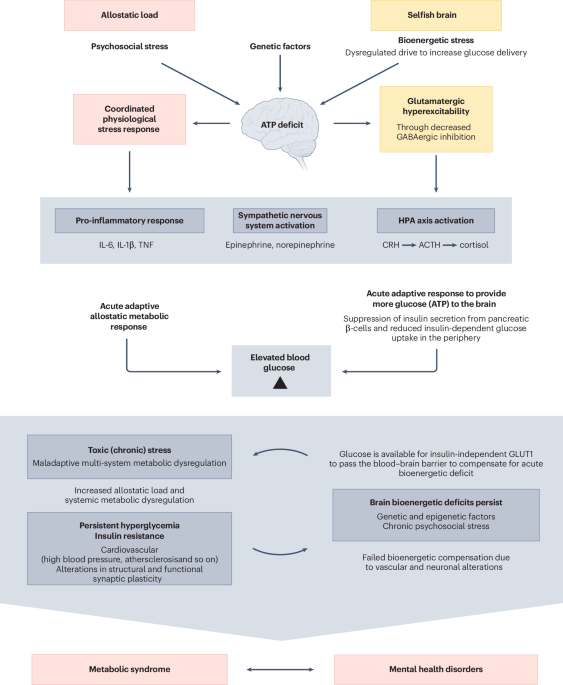
Link Securities | Pharmamar (PHM) announced to the National Securities Market Commission (CNMV) that the Australian Therapeutic Goods Administration (TGA) and Singapore’s Health Sciences Authority (HSA) have granted approval for Zepzelca (lurbinectedin) in combination with atezolizumab (Tecentriq) as a first-line maintenance treatment for adults with advanced small cell lung cancer (ES-SCLC) whose disease has not progressed following first-line induction treatment with atezolizumab, carboplatin and etoposide.